Identifying Authentic Janssen HIV Medicines
Date Last Updated: 12/07/2022
HIV medicines have become a focus of illegal counterfeiting activity in recent years, impacting multiple products and companies in the United States.
Patients, healthcare providers and pharmacists can identify authentic HIV medicines versus a counterfeit form by looking at the tablet. Janssen HIV medicine tablet descriptions are listed below for SYMTUZA® (darunavir/cobicistat/emtricitabine/tenofovir alafenamide), PREZCOBIX® (darunavir/cobicistat), PREZISTA® (darunavir), EDURANT® (rilpivirine) and INTELENCE® (etravirine).
Product information for these medicines can be found on https://www.janssen.com/us/our-products.
SYMTUZA®
FDA-approved SYMTUZA® tablets are yellow to yellowish-brown, capsule-shaped, film-coated tablets debossed with “8121” on one side and “JG” on the other side.

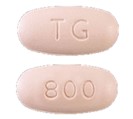
PREZCOBIX®
FDA-approved PREZCOBIX® tablets are pink, oval-shaped, film-coated tablets debossed with “TG” on one side and “800” on the other side.
PREZISTA® 800 mg
FDA-approved PREZISTA® 800 mg tablets are dark red, oval-shaped, film-coated tablets debossed with “T” on one side and “800” on the other side.

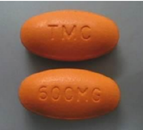
PREZISTA® 600 mg
FDA-approved PREZISTA® 600 mg tablets are orange, oval-shaped, film-coated tablets debossed with “TMC” on one side and “600MG” on the other side.
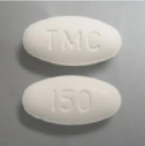
PREZISTA® 150 mg
FDA-approved PREZISTA® 150 mg tablets are white, oval-shaped, film-coated tablets debossed with “150” on one side and “TMC” on the other side.
PREZISTA® 75 mg
FDA-approved PREZISTA® 75 mg tablets are white, caplet-shaped, film-coated tablets debossed with “75” on one side and “TMC” on the other side.

EDURANT®
FDA-approved EDURANT® tablets are white to off-white, film-coated, round, biconvex, 6.4 mm tablets debossed with “TMC” on one side and “25” on the other side.

INTELENCE® 200 mg
FDA-approved INTELENCE® 200 mg tablets are white to off-white, biconvex, oblong tablets debossed with “T200” on one side.

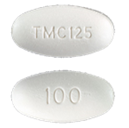
INTELENCE® 100 mg
FDA-approved INTELENCE® 100 mg tablets are white to off-white oval tablets debossed with “TMC125” on one side and “100” on the other side.
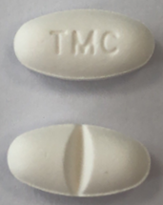
INTELENCE® 25 mg
FDA-approved INTELENCE® 25 mg tablets are white to off-white, oval, scored tablets debossed with “TMC” on one side.
(Pills Are Not Actual Size)
What should I do if I suspect counterfeit HIV medicine?
If tablets in any bottle of medicine do not match the description of the prescribed product, or if the packaging, labeling or inserts accompanying the bottle appear to adulterated, false or altered this should be reported immediately to the FDA’s Office of Criminal Investigations (OCI) at 1-800-551-3989 (http://www.fda.gov/OCI) or Janssen Medical Information at 1-800-JANSSEN (1-800-526-7736), or [email protected], or at https://AskJanssenMedicalInformation.com. If patients identify counterfeit product, they should not take the suspect medicine and should contact their prescriber immediately to ensure they can access authentic HIV medication and adequately continue antiretroviral treatment.
If a patient is experiencing any adverse effects related to Janssen medicines or to the use of counterfeit medicines, they should immediately contact FDA’s MedWatch Program (1-800-FDA-1088) or Janssen Medical Information at 1-800-JANSSEN (1-800-526-7736), or [email protected], or at https://AskJanssenMedicalInformation.com.